Records so that they stand the test of time? How do you make sure that the computers in your
Program ContentCourse Overview:How do you replace paper records with electronic data and make sure the system isĬompletely transparent, robust and tamperproof? And how do you store those electronic data Webinar tutorials which entail self-assessments at the end of each week and gave me invaluable experience It is very well delivered through weekly lectures and It very easy to continue studying even though I found employment 2 months Dr Joe Bradys presentation on the videos and his expertise and help at the webinars was outstanding. Videos supplied were excellent, the notes tied in very efficiently and accurately with the videos. I found the course was run very professionally, The course notes and Their insights and experience into a program that gets you results. They will deliver the most up-to-date course content while blending Who are the Lecturers?All our lecturers continue to work with industry and have years of frontline industry and
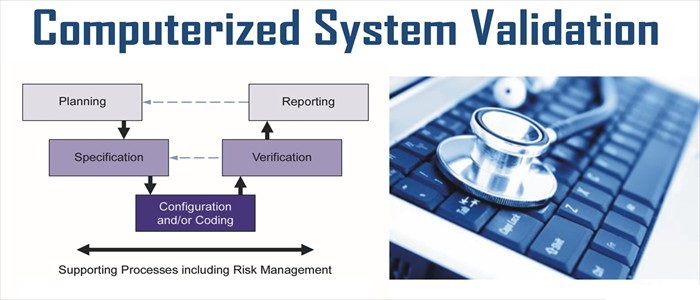
GAMP 5 GUIDELINES HOW TO
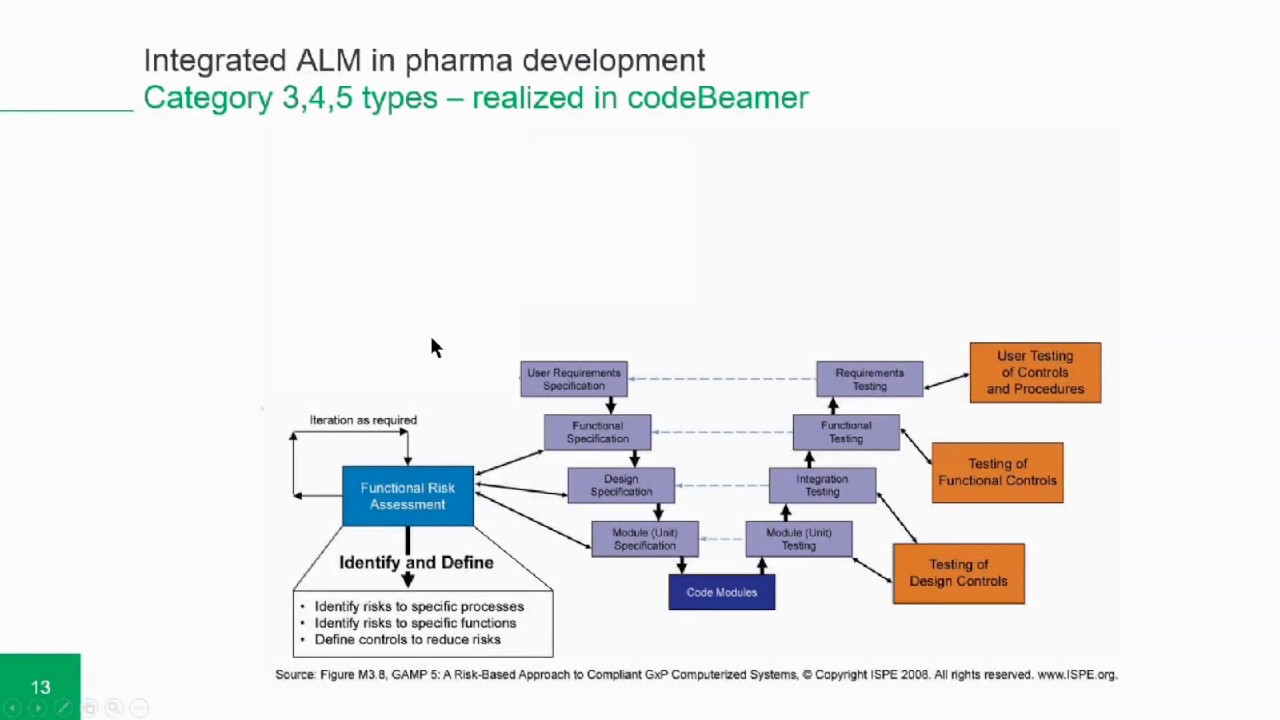
What will you learn?You will learn how to manage electronic data in line with the rules and guidelines of GAMPĥ to ensure that it meets the FDA requirements for manufacturing safe medicines and medical In addition, you will also be requested to regularly post on our discussion board forumĪnd invited to join our LinkedIn Groups of Alumni.
GAMP 5 GUIDELINES FULL
This module culminates in the completion of a full assignment which offers you the opportunity to solidify your knowledge and apply the content in a real world situation. Online learning environment where you can communicate with us, your classmates and Minute) content rich videos, downloadable notes, case studies, and worked examplesĪllowing you to complete the course at your own pace. What can you expect? This module is delivered on our Learning Management System with a mixture of short (10 Pursue or advance their career in the pharmaceutical or medical device manufacturing industry Manufacturing, science, engineering, quality or logistical background and who would like to Program OverviewWho is this module for?GAMP 5 (Good Automated Manufacturing Practice) is suitable for anyone with a
GAMP 5 GUIDELINES UPGRADE
Who are currently in this sector and want to upgrade their skills set. Pharmaceutical or Medical Device Manufacturing Industries or those Ideal for people who want to make a career change into the Get an industry recognised qualification and confirm your knowledgeĭelivered online worldwide on our learning management system withĢ4/7 access. Join the 3,150 people over the last 10 years who have used ourĬourses to build their careers in these industries worldwide. Manufacturing Industries, in the shortest time possible with our WelcomeAccelerate your career in the Pharmaceutical and Medical Device Good Automated Manufacturing Practices (GAMP 5)
